About Us
MedAlliance is a privately-owned medical technology company. It is headquartered in Nyon, Switzerland, with offices in Germany, Singapore, UK and USA.
MedAlliance has been dedicated to developing innovative drug-eluting balloons (DEBs) for patients suffering from life-threatening coronary and peripheral arterial disease. Using breakthrough proprietary technology, MedAlliance has found a unique solution to the challenge of controlled and sustained sirolimus release with SELUTION SLR™.
SELUTION SLR - Breakthrough Technology to Deliver Sustained Limus Release for up to 90 days1
In February 2020 MedAlliance announced the award of its first CE Mark Approval for SELUTION SLR (PTA) in the treatment of peripheral arterial disease and in May 2020 the company was awarded their second CE Mark for SELUTION SLR (PTCA) for the treatment of coronary arterial disease.
SELUTION SLR is the latest generation of drug-eluting balloon technology for coronary and peripheral interventions with sustained limus release. Designed to deliver the same safety and performance as the best-in-class DES technology 2 with the added benefit of “leaving nothing behind”. Learn more...
View more content from MedAlliance on our YouTube channel

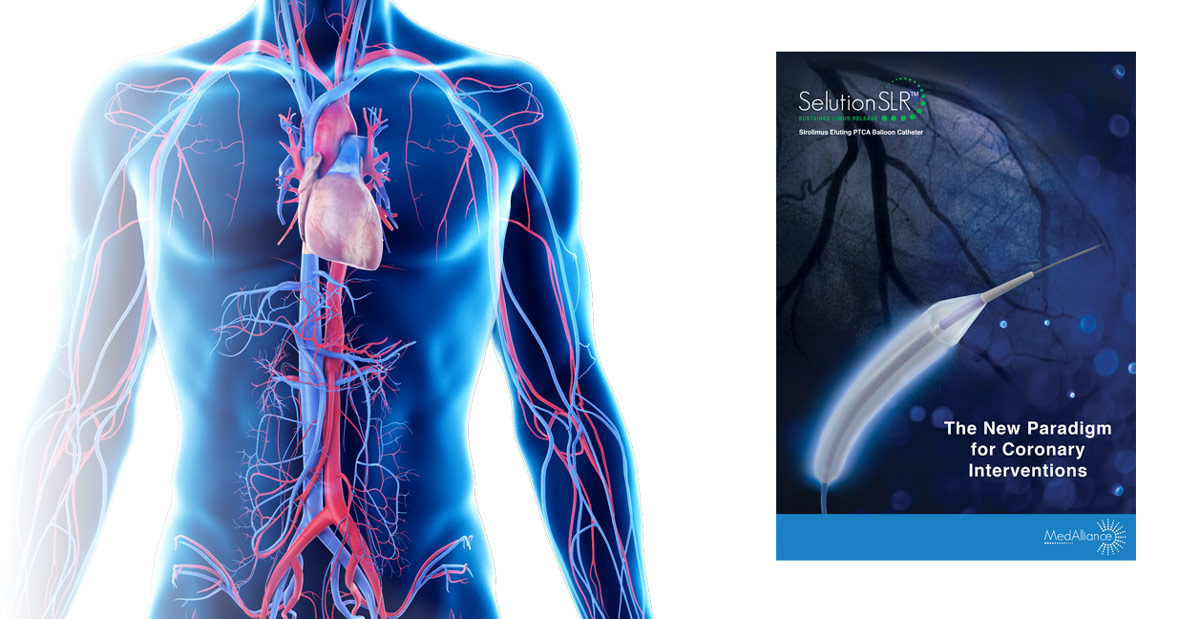
SELUTION SLR 014 Sirolimus Eluting PTCA Balloon Catheter
Intended for use as a Percutaneous Transluminal Coronary Angioplasty (PTCA) balloon catheter to dilate de novo or restenotic coronary lesions, for the purpose of improving myocardial perfusion and decreasing the incidence of restenosis.
SELUTION SLR offers the broadest range of PTCA drug-eluting balloon catheters with balloon diameters ranging from 1.50 mm to 5.00 mm and lengths from 10 mm to 40 mm.
Learn More
Learn more

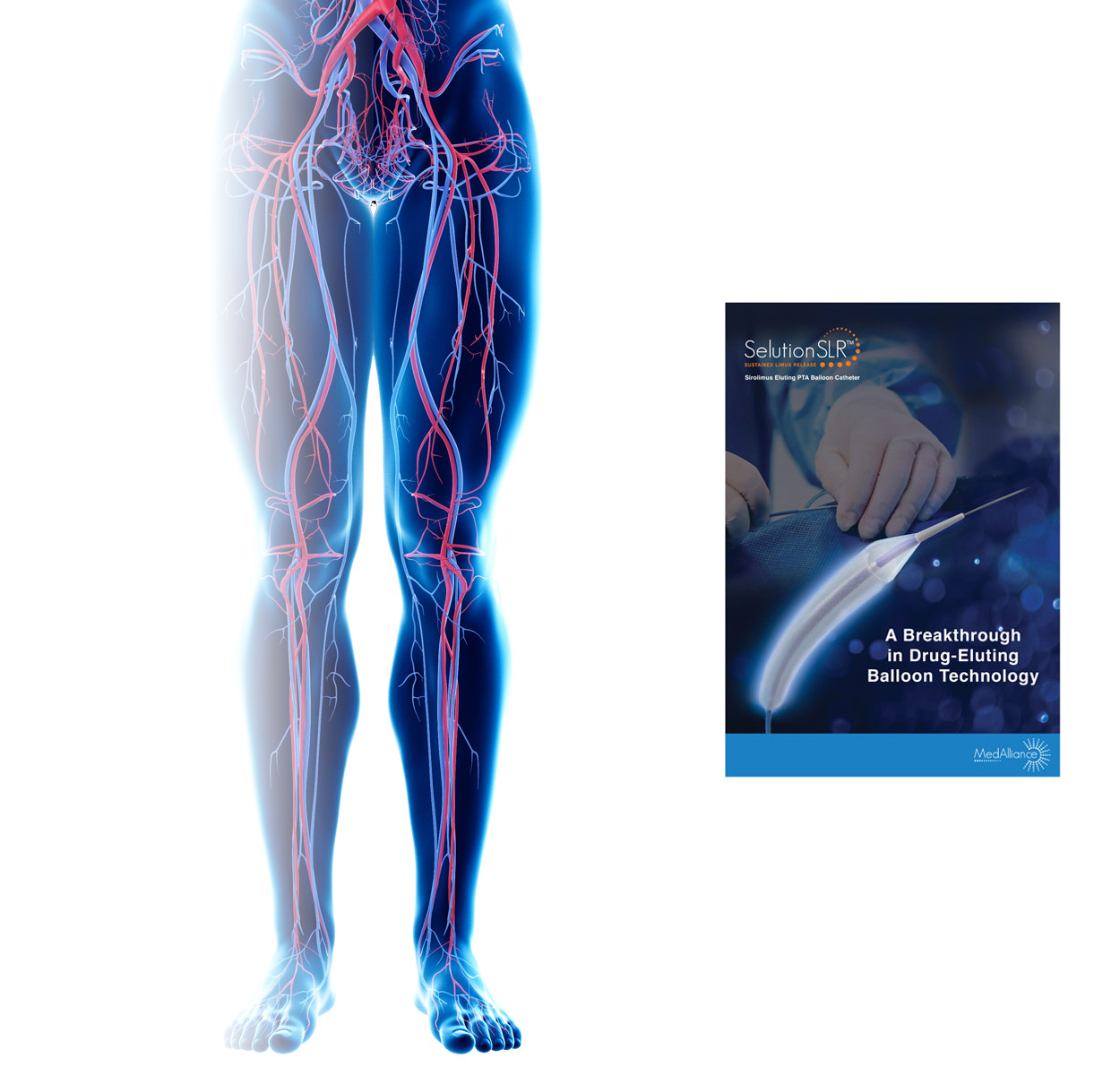
SELUTION SLR 018 Sirolimus Eluting PTA Balloon Catheter
Intended for use as a Percutaneous Transluminal Angioplasty (PTA) balloon catheter to dilate de novo or restenotic vascular lesions, for the purpose of improving limb perfusion and decreasing the incidence of restenosis.
SELUTION SLR offers the broadest range of PTA drug-eluting balloon catheters with balloon diameters ranging from 2.00 mm to 7.00 mm and lengths from 20 mm to 150 mm.
Data References:
- Drug concentration evident in MicroReservoirs and tissue - Data on file at M.A. Med Alliance SA.
- To achieve optimum performance from SELUTION SLR as a possible alternative to DES, follow the Instructions for Use in your implant procedure, and reference “How to use the drug-eluting balloon: recommendations by the German consensus group”, EuroIntervention 2011;7:K125-K128






